PROCOPE Medicals, designer of a new-generation artificial heart, starts preclinical trials in 2024
Nantes, April 15, 2024 – PROCOPE Medicals, a medtech born of a multidisciplinary encounter between engineers and cardiac surgeons, is designing a total artificial heart offering an innovative and inclusive solution for patients suffering from severe heart failure. The startup announces the start of the pre-clinical trial phase in 2024. This marks a further essential milestone in the development of its device, following the proof-of-concept validated by laboratory tests, the numerical simulations carried out to ensure the hemodynamics of the artificial ventricles, and the morphological compatibility tests carried out in 2022 to optimize the size of the prosthesis.
Responding to a global health problem
Every year, 200,000 people wait for a heart transplant. Today, access to heart transplantation is severely limited by the shortage of grafts. There is an urgent need to find a technological alternative to an unmet global need. Since 2018, based on patented pneumatic technology, PROCOPE Medicals has been working with cardiac surgeons at Nantes University Hospital to design a prosthesis that reproduces the pulsatility of the natural heart.

Innovation for heart health
In a market estimated at $16 billion, PROCOPE Medicals intends to make a difference by eliminating the major problems encountered by patients and doctors alike.
encountered by patients and doctors alike: current cardiac prostheses are necessarily connected, via cables, to external equipment through the abdomen, leading to infections. What’s more, because of their size and volume, some devices can only be implanted in individuals with a sufficiently large ribcage, thus excluding a proportion of patients, particularly women.
“Current solutions are not sufficient to alleviate the shortage of heart transplants,” explains CEO and Scientific Director Saïd Chabane.
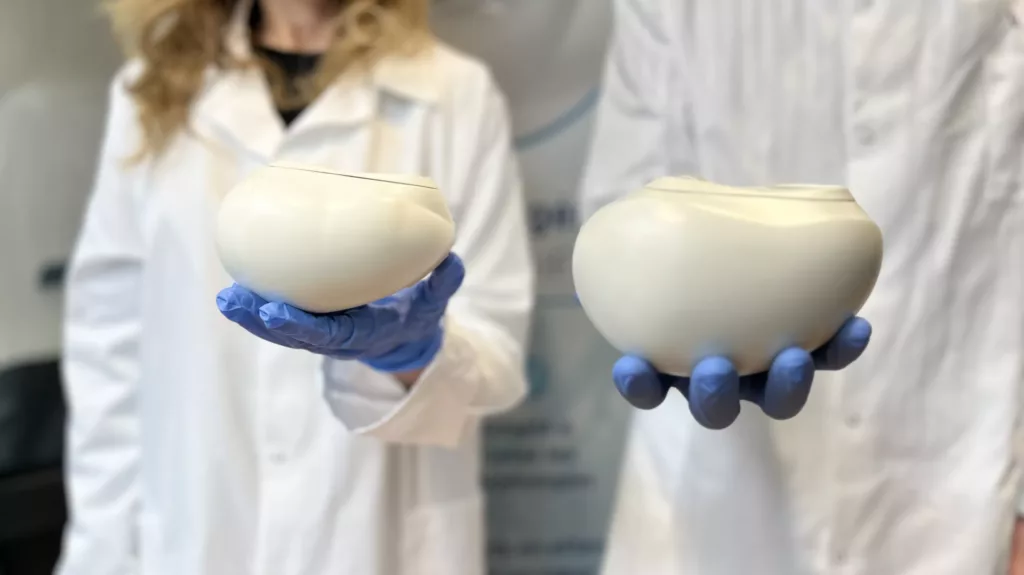
PROCOPE Medicals has designed a solution that sets itself apart from the competition through its innovative vision: the artificial heart has no external wired connections, which are a source of infection, and is available in two volumes to adapt to most morphologies. It reproduces the self-regulated activity and pulsatility of the natural heart. Finally, on-board intelligence enables remote transmission of physiological data to medical teams. The aim is to considerably improve patients’ quality of life and limit the risk of complications.
Pre-clinical trials ; a decisive step forward
In-vivo feasibility tests will be carried out on non-genetically modified pigs, in collaboration with Oniris, the veterinary school in Nantes. The aim will be to confirm the efficacy and safety of the device before moving on to human clinical trials, scheduled for 2027. The team currently comprises 9 talents, and the company aims to increase recruitment to 15 by 2025.
Call for investors
To finance these developments, PROCOPE Medicals will continue to rely on private and public funding. The company has already raised 2.5 million euros, and now intends to raise 9 million euros, in two stages, to finance the preclinical phase (feasibility and animal demonstration). “In 2024, a first stage of financing is being carried out to raise 2 million euros, followed by 7 million euros in 2025, from Venture Capital (VC) and Family Offices to achieve the definitive version of the prosthesis before human trials,” explains Stéphanie Gouraud, Director of Operations and Communications. Financing requirements are estimated at 46 million euros up to market launch in 2030. The support of investors will enable the realization of a project with global impact.
About PROCOPE Medicals
PROCOPE Medicals is a French medtech company based in Nantes, developing an innovative artificial heart that will enable all patients suffering from acute heart failure to regain greater quality of life and autonomy post-surgery, as well as providing practitioners with a reliable and inclusive alternative. The technology, protected by an international patent, eliminates the need for infection-prone transcutaneous wires and makes the device fully implantable. The entire team, engineers and doctors alike, is working to provide the best possible solution to the global problem of the shortage of heart transplants.
Press contact :
Stéphanie Gouraud, 06 83 24 81 41, sgouraud@procope-medicals.com
Marie Dallibert, mdallibert@procope-medicals.com
Website : https://procope-medicals.com/
